-
Who we are
About Us
As one of APAC's leading filtration specialist, BHF design, fabricate, install and service liquid and gas filtration and water purification systems that improve critical processes for the wine, food and beverage, healthcare and biotechnology, water treatment, mining and resources industries.
Meet the Team
Meet our diverse group of industry specialists whose vast experience and innovative thinking keep us at the forefront of the filtration and purification industry.
Sustainability and Governance
We are dedicated to sustainable and responsible operations in our own business as well as supporting it in client industries. Our commitment to Environmental stewardship, Safety, and Governance (ESG) is integrated into everything we do
-
What We Do
Oenological additives








Service & Maintenance
Our national team of dedicated specialist technicians offer both contracted and on-demand customer support, calibration, remote monitoring, upgrades and preventative maintenance, as well as parts-stock management programs.
Site Audit
Our experts are ready to assess your current system performance and advise on opportunities to improve efficiency and performance whilst reducing operating costs.
-
Industries
Food and Beverage









Water Treatment and Wastewater













-
Our Projects & Media
Projects
Browse some of the innovative systems we’ve created and installed across various industries. Each case study demonstrates how partnering with BHF Technologies resulted in performance improvements.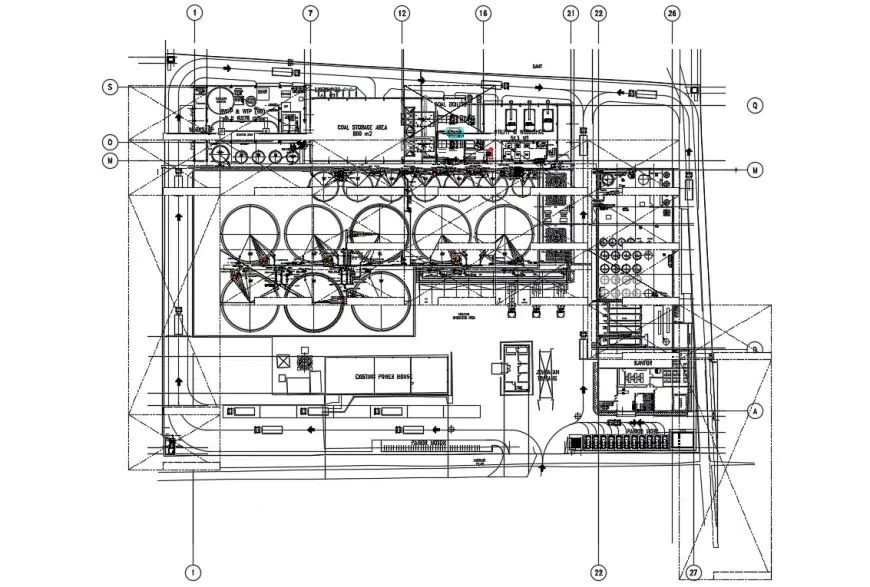
Case Studies
Browse some of the innovative systems we’ve created and installed across various industries. Each case study demonstrates how partnering with BHF Technologies resulted in performance improvements.
Videos
Dive into our collection of videos, demonstrating our filtration expertise. This video repository is designed to showcase the application and utility of our advanced filtration products and systems across various industries. From detailed process demonstrations to practical tutorials on equipment operation and maintenance, each video is crafted to provide you with valuable insights and enhance your understanding of our innovative solutions.

-
Resources
Downloads
Our library of downloadable PDFs includes a diverse array of technical documents designed to support every aspect of your filtration and purification needs. Our wide-ranging and accessible technical resources have been curated to provide you with thorough insights and critical data that will enhance your operational knowledge and ensure compliance and high performance of your filtration and purification applications.

-
News & Blog
Blog
New technologies, new partnerships, operational breakthroughs … you’ll find all the exciting developments plus a range of interesting thought pieces here.
Events
As a leader in filtration and water purification across APAC, BHF actively participates in a wide range of industry events — from technical workshops and training sessions to major conferences. These engagements keep us at the forefront of innovation and sustainability, while allowing us to share our expertise, collaborate with industry peers, and support knowledge … Continued
- Contact Us














