Coinoculation of Yeast & Bacteria
August 26, 2024
In the most basic form of winemaking grapes are crushed and allowed to commence primary (alcoholic; AF) and secondary (malolactic; MLF) fermentation according to the dictates of nature. Whilst this is a harmonic concept in ideology, the reality is that “wild” primary and secondary ferments can generate both significant process and wine quality problems for a winemaker, if indeed one or both ferments finish at all. Most winery business models do not cater for the intrinsic potential process unreliability and wine quality risks associated with this approach, and so winemakers have turned more frequently to inoculated primary and secondary ferments.
The inoculation of a fermentation with yeast has been accepted practice for decades, but the use of bacterial starter cultures is a more recent methodology. Initially, MLF cultures were added sequentially, only after AF was complete. More recently, this practice has evolved to constitute a coinoculation, whereby the bacterial culture is added whilst the yeast culture is active. This article explores the manifold advantages of employing coinoculation, in terms of winemaking process, quality and economics.
Why inoculate with selected bacteria at all?
Winemakers often ask this question. Certainly, MLF can occur spontaneously, but there can be problems in the resulting wine even if this occurs. Indigenous bacteria typically produce biogenic amines through the action of the bacterial decarboxylase enzymes on the precursor amino acids. These can generate allergenic reactions in some consumers, usually manifesting as headaches or skin irritation. Other biogenic amines have unpleasant odours, such as the aptly-named putrescine and cadaverine, which diminishes wine quality.
Commercial bacteria (LAB; lactic acid bacteria, most commonly Oenococcus oeni in winemaking) are typically selected for several characteristics: high alcohol/SO2 and low pH tolerance, resistance to low temperatures and, more recently, minimal biogenic amine production (Figure 1).
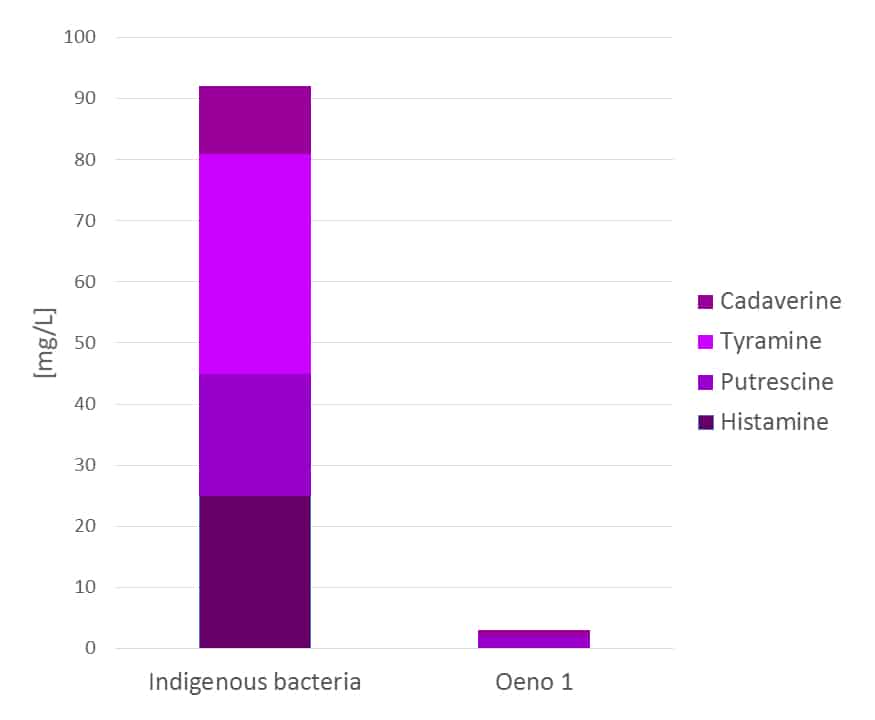
Figure 1: The biogenic amine production of indigenous bacteria vs. Oeno 1, a commercially selected strain. The wine used was a 2009 Medoc Merlot, 14.8 % alc., pH 3.52/TA 3.85 g/L (H2SO4), TSO2 36 ppm.
The usual latency period between AF and wild MLF allows an open window of opportunity for spoilage microorganisms to invade due to the absence of SO2, chiefly Brettanomyces. Wild MLF fermentation kinetics are often poor when compared with selected bacteria (Figure 2), which can become a critical (and expensive) factor when either tank space/process/labour time or ambient temperatures become control points, and the cost associated with heating wine can be significant. Some wild malolactic fermentations do not complete at all (Figure 2).
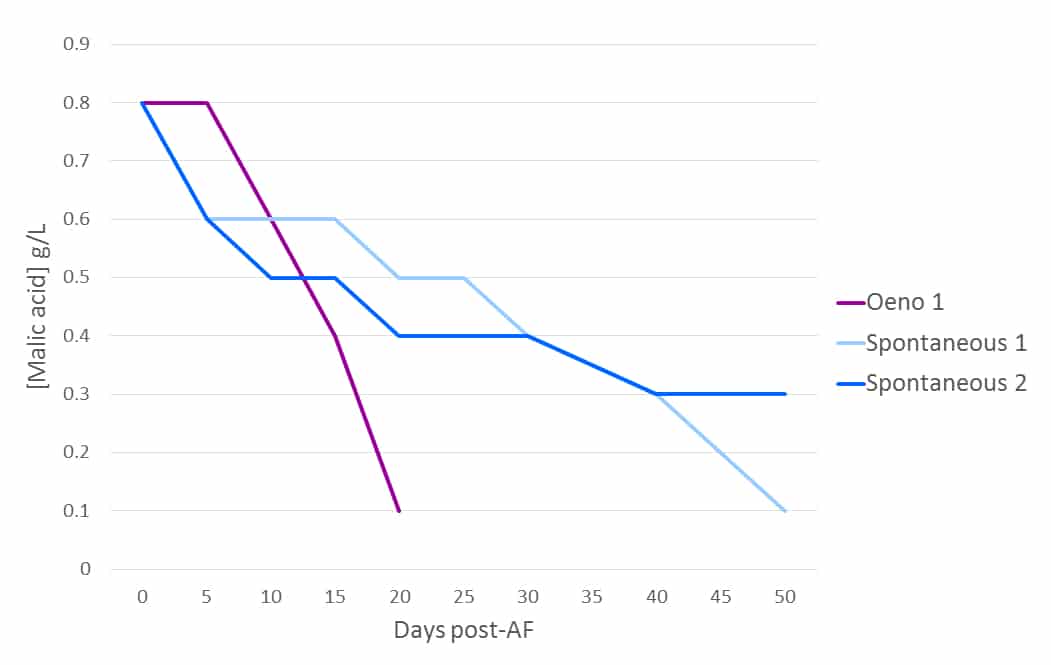
Figure 2: The fermentation kinetics of indigenous vs. Oeno 1. Oeno 1 was inoculated during pressing in this example. The wine used was a 2009 Medoc Merlot, 14.8 % alc., pH 3.52/TA 3.85 g/L (H2SO4), TSO2 36 ppm.
LAB also vary in their production of diacetyl. At low levels diacetyl contributes a butter/butterscotch-like character to wine, but at higher levels this turns to a more chemical aroma. Diacetyl is considered by many winemakers to be undesirable, and to detract from fruit expression, hence most commercial LAB strains are selected with minimal diacetyl production in mind. Indigenous LAB malolactic fermentations thus typically produce higher levels of diacetyl. Since diacetyl is a product of citric acid metabolism, which typically occurs only in the absence of malic acid, to minimise diacetyl production a commercial LAB inoculum should be used, the malolactic fermentation closely monitored, and the wine sulfited as soon as all malic acid has been consumed. In the event that some diacetyl is desired, simply hold off on sulfiting at the end of MLF for around 2-10 days, or until sufficient diacetyl has been produced.
Figure 3 shows the malic and citric acid metabolism profiles of Oeno 1. Note that citric acid metabolism only occurs after malic acid depletion (scale at left), indicated on day 8, with the subsequent production of diacetyl (scale at right).
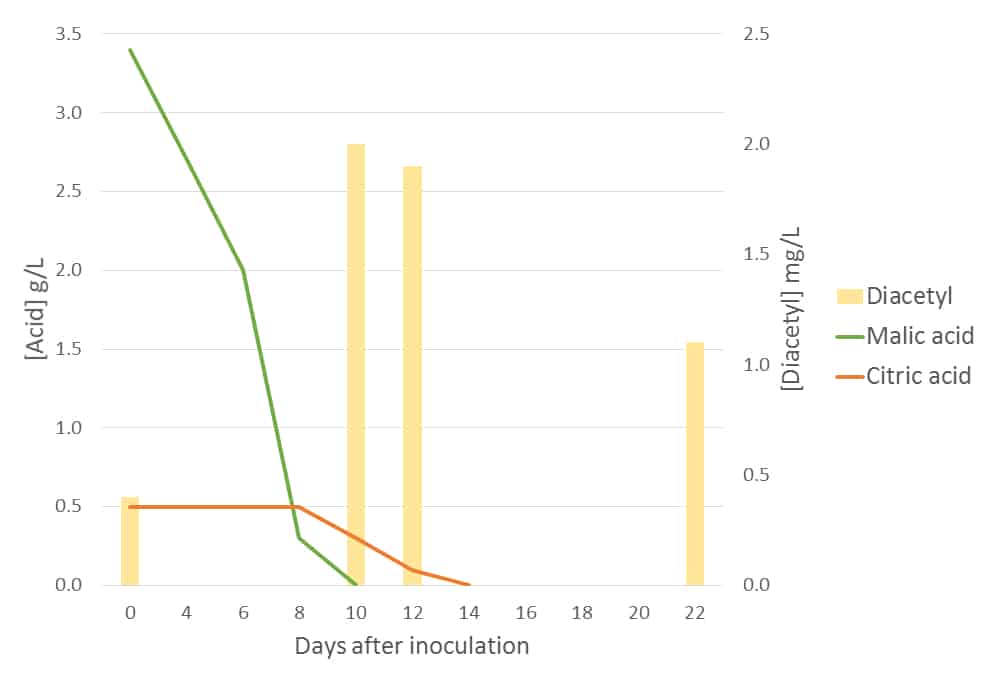
Figure 3: The metabolism of malic and citric acids, and subsequent production of diacetyl, by commercial LAB Oeno 1. Diacetyl concentration was measured on days 0 (inoculation), 10, 12 and 22.
Another reason to inoculate with selected LAB is the production of free cinnamic acids by indigenous bacteria, which are Brettanomyces substrates. Some strains of O. oeni possess cinnamyl esterase activity, which cleaves the (mainly tartaric) ester precursor cinnamic acid form, allowing Brettanomyces to then produce vinyl (chemical/medicinal odours) and then ethyl (horse/band-aid odours) phenols. These compounds are well known, and are not pleasant. The main marker compounds here are 4-ethylphenol (4EG) and 4-ethylguaiacol (4EG). This also relates to the use of non-purified enzymes (Bowyer & Mennesson, 2020) and POF+ yeast (Bowyer et al, 2017; Figure 4) in the earlier stages if winemaking.
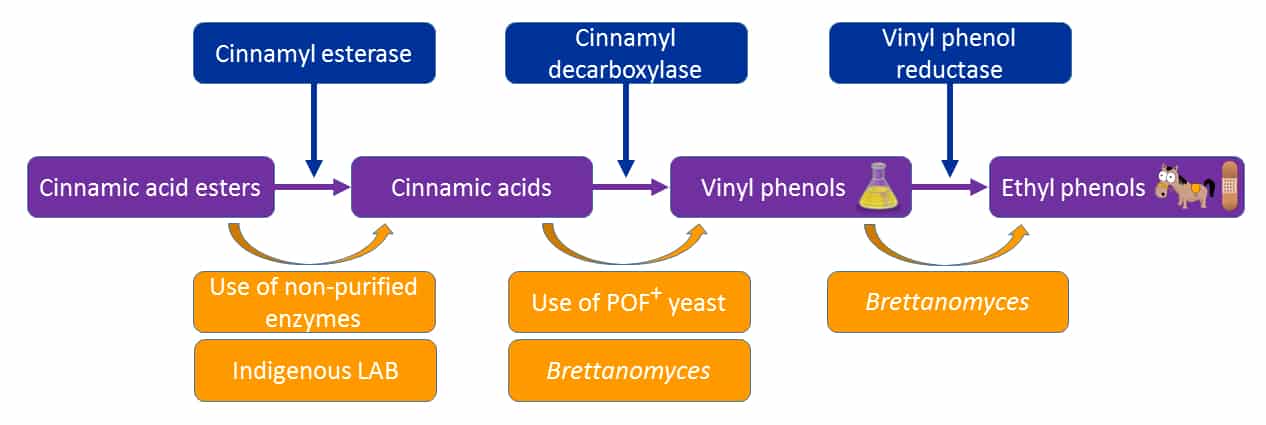
Figure 4. The contributory pathways through which Brettanomyces can generate ethylphenols in wine. Enzymes are shown in blue, substrates and metabolites in purple, and process/organisms in gold.
Selected bacterial strains are also more resistant to stress factors, such as low pH and inhibitory compounds like chitosan and SO2 (Figure 5). This is important, as indigenous bacteria are typically very sensitive to these treatments. Chitosan (Bowyer et al, 2019) is a recent Brettanomyces management tool, and a bacterium that displays resistance to this treatment is extremely useful.
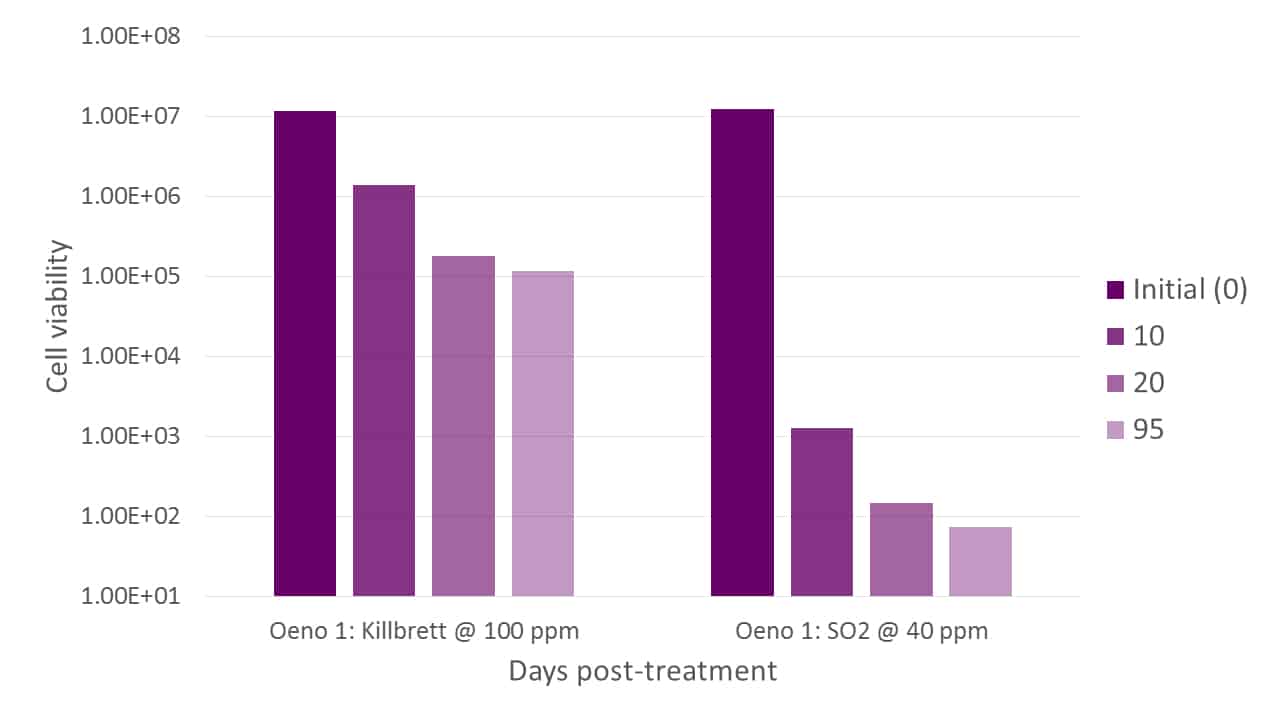
Figure 5. The resistance of Oeno 1 to the stress factors chitosan (KillbrettTM, left) and SO2 (right). Cell viability is shown on addition of the stress factor, then after 10, 20 and 95 days.
The malolactic fermentability index
This is an estimation of the level of difficulty that can be expected for MLF to occur in a given wine. The index is derived from an amalgam of the more critical parameters associated with this determination (Table 1).
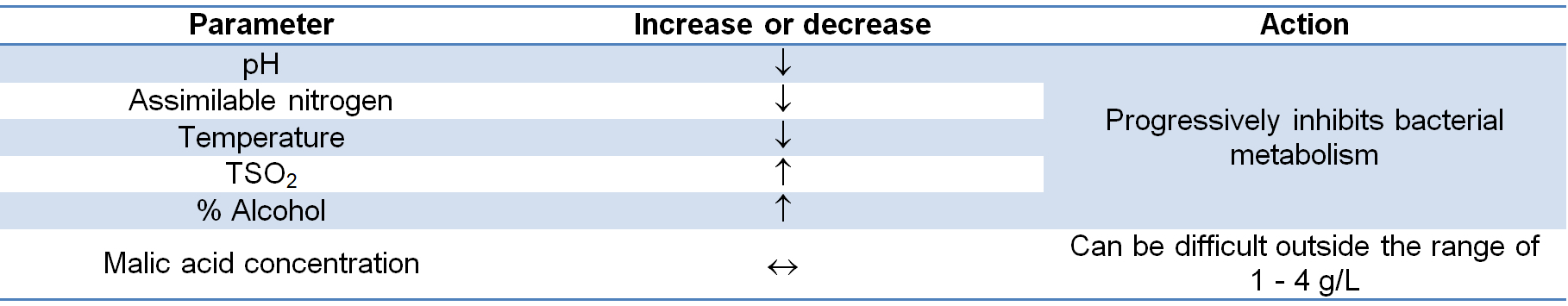
Table 1: Factors used to determine the malolactic fermentability of a wine.
Why coinoculate, rather than sequentially inoculate?
In short, for increased process speed, fermentation reliability and superior wine quality.
Indigenous LAB typically have an unreliable and unpredictable growth phase, and as a rule of thumb express little activity until a population threshold of around 106 cells/mL is reached. Additionally, they tend to be more susceptible to environmental pressures than selected strains. During the growth phase the wine is non-sulfited, and so the opportunity for spoilage organisms to move in is significant. Inoculation with a commercial LAB strain like Oeno 1 circumvents the bacterial growth phase, allowing secondary fermentation to commence more rapidly. There is a caveat, however: the population threshold must be achieved on inoculation, hence there must be strict adherence to the inoculum dosage. Given the cost of bacterial cultures, there is a temptation to spread the inoculum over a larger wine volume than is stipulated on the packaging. This typically results in an inoculation below the population threshold, which in turn generates a growth phase in the commercial bacteria, somewhat negating the advantages offered by the inoculum in the first place. Best practice is to stick to the recommended application dosage.
Ethanal (acetaldehyde) is a strong binding agent for SO2, and thus leads to elevated TSO2 in the final wine. The ethanal concentration is typically lower for coinoculated wines than for those that are sequentially inoculated. The lower ethanal concentration in coinoculated wines produces less bound/total SO2, and so overall sulfiting is more effective and lower TSO2 results.
Coinoculation: early, late or not at all?
There are two types of coinoculation: early and late. Early coinoculation is when the inoculum is added just after the commencement of AF, typically on day 2 when there is an active cover of CO2, which is important to avoid the production of acetic rather than lactic acid (Bartle et al, 2019). Late coinoculation is when the bacterial inoculum is added towards the end of AF, typically at pressing. Each method has pros and cons, some of which are outlined in Table 2.
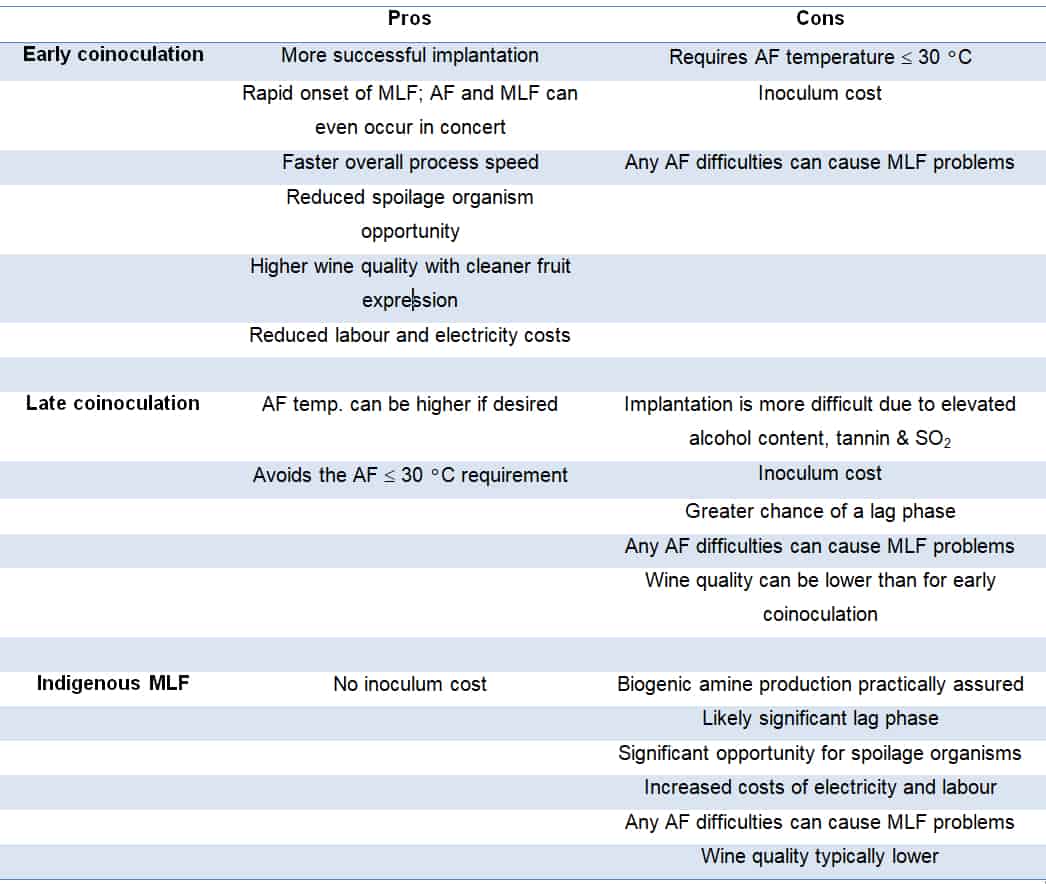
Table 2: Some pros and cons of early vs. late coinoculation.
Yeast and bacterium compatibility
It is import to understand that a certain level of yeast and bacterium compatibility is required for the successful employment of coinoculation. The two organisms are being asked to function essentially as a couple. An expectation that any yeast/bacterium combination can be used for coinoculation is likely to end in disaster. Several factors should be considered when seeking an effective strain combination (Bartle et al, 2019), some of which are discussed below.
Yeast fatty acid production
The production of medium-chain fatty acids varies according to yeast strain, and also increases under stressful conditions during AF. The free forms of these fatty acids are inhibitory to LAB due to their impact on intracellular pH and subsequent trans-membrane function. Yeast strains vary considerably in their production of medium-chain fatty acids (Figure 6) and can produce higher concentrations of these compounds under stress, such as at high alcohol levels, high temperature and when nitrogen and/or minerals and enzymatic cofactors are low or absent.
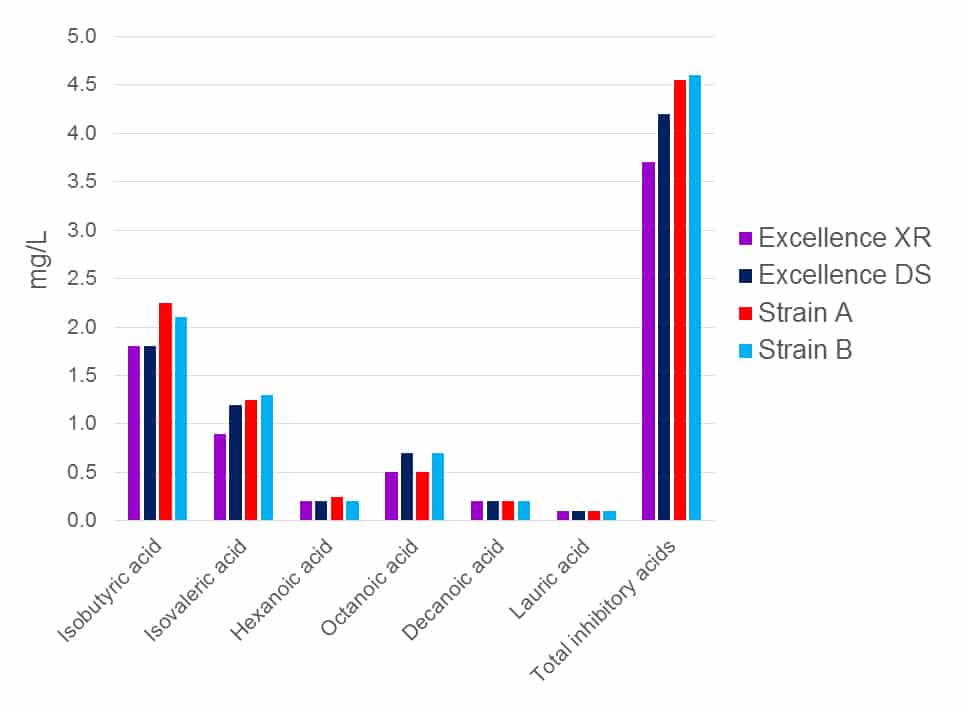
Figure 6. MLF-inhibitory fatty acid production of Excellence XR, Excellence DS and two other commercial yeast strains. Lower overall fatty acid levels lead to faster MLF.
Nutrient consumption
The usage of high nutrient demand yeast is less likely to facilitate a successful coinoculation, unless the proactive management of fermentation nutrient is undertaken. Since bacteria cannot typically metabolise inorganic nitrogen, a complex organic (ie no–DAP) nutrient is advised for primary fermentation.
Yeast SO2 production
All yeast produce some SO2 during AF. Strains that produce more SO2 are less likely to afford a successful coinoculation, as bacteria are especially sensitive to SO2. Further, this situation is exacerbated when juice/must pH is lower, due to the increased proportion of molecular SO2.
Yeast fermentation kinetics
Excessive vigour in the primary fermentation can lead to high must temperatures if winery refrigeration is inadequate. Above 25ºC bacteria become increasingly uncomfortable, and significantly beyond 30ºC cell necrosis can occur. In such cases the inoculum can be severely depleted or destroyed.
Case study: Coonawarra Cabernet sauvignon 2020
In the vintage of 2020 a trial was conducted at a Coonawarra winery to compare the validity of a different approach to red wine production, utilising early coinoculation. Normal practice at the winery is to inoculate the must with Strain B yeast, mostly complete primary fermentation and then inoculate with Strain V bacteria at the press tray with some sugar remaining – a late coinoculation. Against this protocol, Excellence XR yeast was rehydrated using Oenostim rehydration nutrient and the Oeno 1 bacterium was added on day 2 of primary fermentation – an early coinoculation. The picking of fruit occurred a day apart between protocols for harvest logistics reasons. Both primary fermentations were run between 19-22 ºC.
Certainly there are some variables in this comparison, but it was simply a case of challenging the status quo modus operandi with a different approach to evaluate any significant differences in both primary and secondary fermentation kinetics and/or wine outcomes in a global manner.
Plotting the primary and secondary fermentation kinetics for both modalities (Figure 7) delivers some very interesting results.
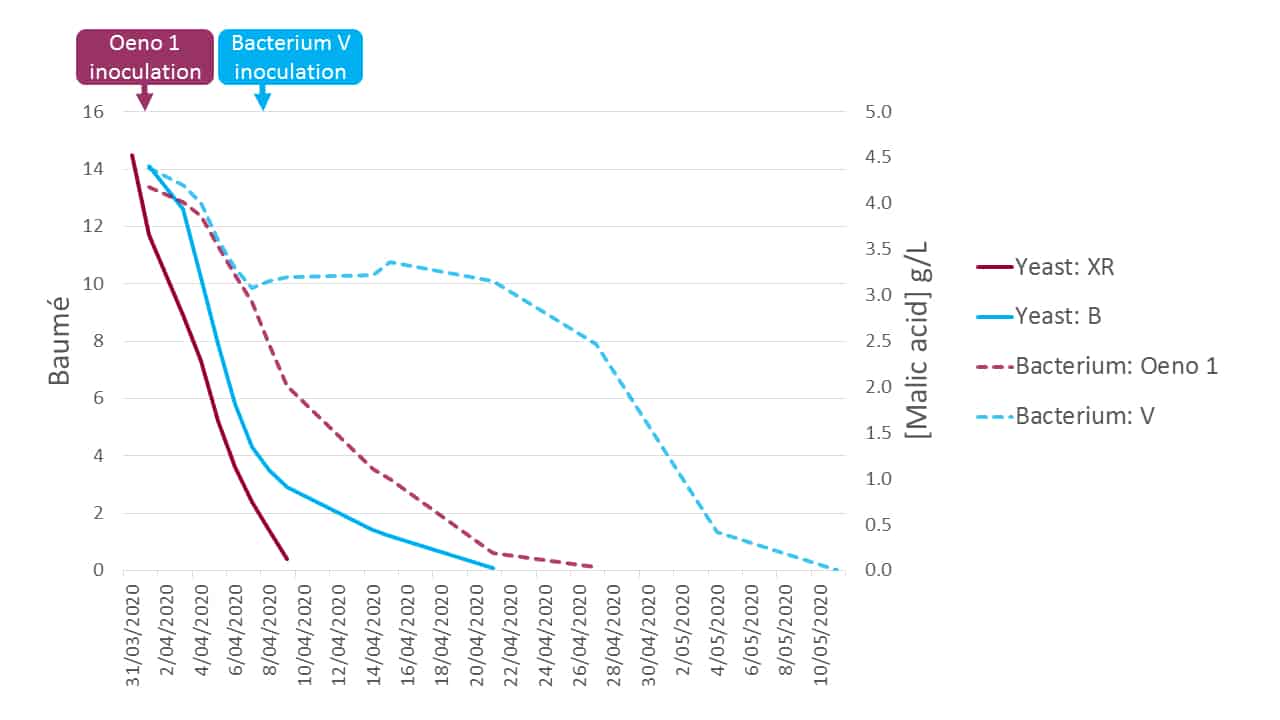
Figure 7. The primary and secondary fermentation kinetics of both early and late coinoculation modalities in a 2020 Coonawarra Cabernet sauvignon protocol comparison.
Strain B yeast/Strain V bacterium
Strain B yeast commenced fermentation well, but struggled at pressing and was sluggish thereafter, eventually completing primary fermentation around 3 weeks after inoculation. Interestingly, malic acid depletion commenced prior to the Strain V bacterial inoculation, with around 1 g of malic acid metabolised, suggesting that either Strain B yeast was consuming it or there was some initial indigenous LAB activity. Strain V bacteria were introduced at pressing at 2.9 Bé and a significant lag phase of some 2 weeks was observed before MLF progressed. Once initiated, MLF completed around 5 weeks after bacterial inoculation.
XR yeast/Oeno 1 bacterium
The fermentation data clearly indicate that both primary and secondary fermentations were progressing in concert. XR completed primary fermentation around 10 days after inoculation. Further, Oeno 1 commenced malolactic fermentation essentially immediately, which is part due to the vigour of the bacterial strain, its method of commercial preparation and also the relatively comfortable physical environment that it was introduced to on day 2 of the primary fermentation. By the time the primary fermentation with XR yeast was complete, the secondary fermentation was about 50 % complete. Importantly, this means that there was no gap in the microbiological niche, and hence the opportunity for spoilage organisms to ingress was minimised. MLF completed within 2 weeks of the primary fermentation.
Sensory differences
On top of the kinetic differences between the modalities compared, there were significant sensory differences between the wines. After 2 months in barrel, the XR/Oeno 1 wine was dominated by primary varietal fruit aromatics of cassis and blackberry, and the tannin profile was very full and soft, a result of the polysaccharide production of XR yeast. In contrast, the Strain B/V wine showed less primary fruit and hints of ethyl acetate, no doubt a result of the difficult primary and secondary fermentations. Additionally, the Strain B/V wine showed noticeable cineole (eucalyptol), which was not apparent in the XR/Oeno 1 wine, although the origin of this difference is as yet unclear. It may simply be that the increased primary fruit evident in the XR/Oeno 1 wine suppressed the cineole aroma in that wine.
Further chemical and sensory studies will be conducted on this wine pair in an effort to explore this aspect of the trial.
Cost of MLF inoculation
A common argument against the use of commercial LAB is the cost of the inoculum. Whilst this can seem to be high when purchasing a sachet of freeze dried bacteria, aside from the previously discussed qualitative and kinetic advantages that a selected bacterial strain offers, there are typically significant logistics and on-cost gains with this approach, encompassing reduced costs in electricity, winery labour, and wine analysis. The electricity savings gained by not having to heat wines in tank or barrel through the onset of colder weather, often required with indigenous MLF, constitutes a strong economic advantage in itself. Wines that have completed MLF can be sulfited earlier, and thereafter require minimal attention, significantly reducing winery labour costs. The continuing chemical analyses required for slow MLF in multiple barrels also represents a significant winery cost. All of these less transparent costs associated with indigenous MLF should be factored in when examining the price of commercial LAB products.
Summary
Inoculation with a selected bacterial strain can deliver significant benefits to both wine quality and the winemaking process on several levels. Winemakers seeking to maximise wine quality and/or increase process efficiency should consider MLF inoculation. Further benefits can be obtained using early rather than late coinoculation or sequential inoculation, but this approach mandates that the yeast and bacterium chosen constitute a synergistic couple. Seeking to utilise early coinoculation with incompatible yeast and bacteria is likely to end in disaster.
Excellence XR and Oeno 1 have proven themselves to constitute a superbly synergistic couple for over a decade. Provided some simple caveats are observed, and complimentary yeast and bacterial couples are used, early coinoculation can represent a significant improvement over existing winery practices and can increase wine quality.
By Paul K. Bowyer and Arnaud Mennesson
BHF wishes to thank The Australian and New Zealand Grapegrower and Winemaker for permission to reproduce this article. Subscription information can be found here.